 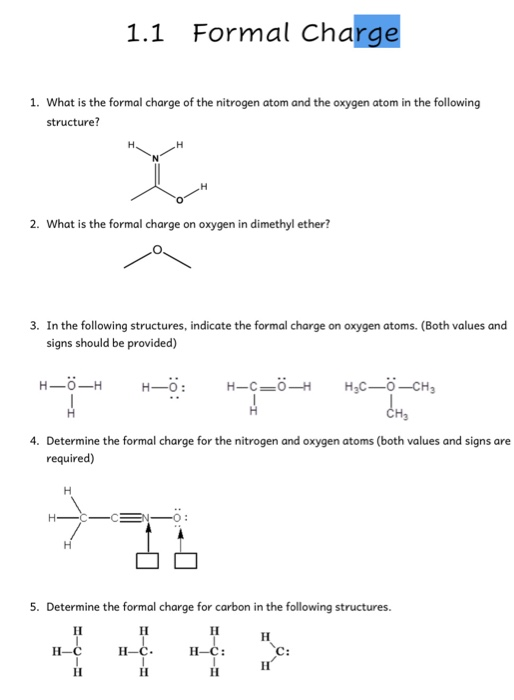
The 10/20 reaction of O 2 + in O 2 was also observed to depend on excited O 2 + ions. First off, dry air is 78 percent nitrogen, 21 percent oxygen, and 1 percent. Nitrogen has 5 valence electrons, this atom has 6 bonded electrons (a. The charge on an atom is related to its valence electrons or oxidation state. You can use this chart to predict whether or not an atom can bond with another atom. The 10/01 collisions of N + in N 2 and O + in O 2 exhibited an increase in cross section as metastables were added to the parent ion beam. Dealerships and tire shops often charge owners to fill tires with nitrogen. The top structure: First, we calculate the formal charge of the nitrogen on the left. Updated on JThis is a chart of the most common charges for atoms of the chemical elements. Also, the reported transfer of the ν = 3 level of the B to the ν = 14 level of the A 2Π u was found.The cross section for 10/01 charged exchange of N 2 + in N 2 exhibited a marked decrease as excited-state ions diluted the beam. From the collision-induced dissociation of N 2 +, the A 2Π u and the states have been identified. In order that an ion reach the collision region, it had to have a mean lifetime greater than 3 μsec.The a 4Π u and b 4Σ g excited states of O 2 + were identified in the collision Higher states of O 2 +, which have not as yet been identified spectroscopically, were found in the collision The thresholds of these new states are 23.9, 27.9, 31.3, and 34.1 eV with an uncertainty ☐.2 eV. In every case studied, cross sections depended markedly upon the presence in the ion beam of ions in metastable or long-lived radiative states. (4.3.1) formal charge valence shell electrons (free atom) lone pair electrons 1 2 bonding electrons We can double-check formal charge calculations by determining the sum of the formal charges for the whole structure. 
curves of parent and product ions have been applied to studies of collision-induced dissociation and charge exchange of oxygen and nitrogen in their parent gas. The mass spectrometer techniques of Aston banding and of comparing I.P. The ability to attract the electrons in bonds is called electronegativity, and since it derives from the same effect as the effective nuclear charge and atomic. 
The role of some excited ions in laboratory ion–molecule reactions has been investigated, and their possible importance in the upper atmosphere considered. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
