 
Radiocarbon dating has allowed key transitions in prehistory to be dated, such as the end of the last ice age, and the beginning of the Neolithic and Bronze Age in different regions. Histories of archaeology often refer to its impact as the "radiocarbon revolution". In addition to permitting more accurate dating within archaeological sites than previous methods, it allows comparison of dates of events across great distances. The development of radiocarbon dating has had a profound impact on archaeology. More recently, accelerator mass spectrometry has become the method of choice it counts all the 14Ĭ atoms in the sample and not just the few that happen to decay during the measurements it can therefore be used with much smaller samples (as small as individual plant seeds), and gives results much more quickly. Measurement of radiocarbon was originally done with beta-counting devices, which counted the amount of beta radiation emitted by decaying 14Ĭ atoms in a sample. Conversely, nuclear testing increased the amount of 14Ĭ in the atmosphere, which reached a maximum in about 1965 of almost double the amount present in the atmosphere prior to nuclear testing. 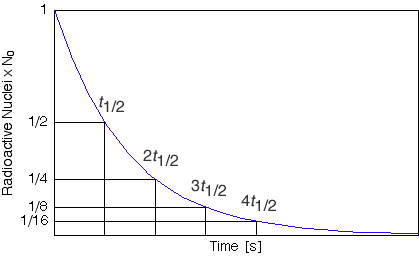
As a result, beginning in the late 19th century, there was a noticeable drop in the proportion of 14Ĭ in the atmosphere as the carbon dioxide generated from burning fossil fuels began to accumulate. Additional complications come from the burning of fossil fuels such as coal and oil, and from the above-ground nuclear tests performed in the 1950s and 1960s.īecause the time it takes to convert biological materials to fossil fuels is substantially longer than the time it takes for its 14Ĭ to decay below detectable levels, fossil fuels contain almost no 14Ĭ. Other corrections must be made to account for the proportion of 14Ĭ in different types of organisms (fractionation), and the varying levels of 14Ĭ throughout the biosphere (reservoir effects). The resulting data, in the form of a calibration curve, is now used to convert a given measurement of radiocarbon in a sample into an estimate of the sample's calendar age. Research has been ongoing since the 1960s to determine what the proportion of 14Ĭ in the atmosphere has been over the past 50,000 years. In 1960, Libby received the Nobel Prize in Chemistry for his work. The older a sample is, the less 14Ĭ there is to be detected, and because the half-life of 14Ĭ (the period of time after which half of a given sample will have decayed) is about 5,730 years, the oldest dates that can be reliably measured by this process date to approximately 50,000 years ago (in this interval about 99.8% of the 14Ĭ will have decayed), although special preparation methods occasionally make an accurate analysis of older samples possible. Measuring the proportion of 14Ĭ in a sample from a dead plant or animal, such as a piece of wood or a fragment of bone, provides information that can be used to calculate when the animal or plant died. When the animal or plant dies, it stops exchanging carbon with its environment, and thereafter the amount of 14Ĭ it contains begins to decrease as the 14Ĭ undergoes radioactive decay. The resulting 14Ĭ combines with atmospheric oxygen to form radioactive carbon dioxide, which is incorporated into plants by photosynthesis animals then acquire 14Ĭ by eating the plants. It is based on the fact that radiocarbon ( 14Ĭ) is constantly being created in the Earth's atmosphere by the interaction of cosmic rays with atmospheric nitrogen. The method was developed in the late 1940s at the University of Chicago by Willard Libby. 
Radiocarbon dating (also referred to as carbon dating or carbon-14 dating) is a method for determining the age of an object containing organic material by using the properties of radiocarbon, a radioactive isotope of carbon. 
Radiocarbon dating helped verify the authenticity of the Dead Sea scrolls. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
